News
News
December 2022
New Alexander von Humboldt Fellow in Inorganic Chemistry
Dr. Oliver Townrow (University of Oxford) has been active since the 1st December 2022 as an Alexander von Humboldt postdoctoral fellow at the Chair of Inorganic and Organometallic Chemistry led by Prof. Dr. Sjoerd Harder. In the coming two years, Dr. Townrow will work on the activation of metals by Metal-Vapour-Synthesis, an area that is currently one of the focal research areas in the Harder group. A major goal of the project is the isolation of complexes with metals in unusually low oxidation states. The extreme reactivity of such complexes can be exploited in the activation of very stable and mostly inert molecules, for example N2 or CO2, and could also lead to conceptually novel catalysts.
April 2022
Dr. Neha Patel erhält Marie-Skłodowska-Curie Postdoc-Stipendium
Dr. Neha Patel ist mit einem Marie-Skłodowska-Curie Postdoc-Stipendium von der Europäischen Kommission ausgezeichnet worden. Dr. Patel ist seit Juli 2021 Postdoktorandin am Lehrstuhl für Anorganische und Metallorganische Chemie (Prof. Sjoerd Harder Ph.D.) und beschäftigt sich im Projekt NITRO EARTH mit der bisher kaum untersuchten Reduktion von Stickstoffverbindungen mit Erdalkalimetallkomplexen. Der Stickstoff-Kreislauf ist entscheidend für die Ökosysteme der Erde.
Frau Patel wird ihre bisherigen Arbeiten an Stickstoff, Stickstoffverbindungen mit niedriger Oxidationsstufe und Erdalkalimetallchemie in der Arbeitsgruppe von Prof. Harder, einer der weltweit führenden Forschungsgruppen in diesem Fachbereich, fortführen. Bei den gemeinsamen Forschungsarbeiten werden modernste Methoden und Techniken beim Umgang mit luftempfindlichen Verbindungen angewendet.
Dr. Patel hat an der Dr. Hari Singh Gour University (Sagar University) in Sagar, Indien Pharmazie studiert und 2018 am Department of Medicinal Chemistry am NIPER S.A.S. Nagar (National Institute of Pharmaceutical Education and Research) promoviert. Anschließend forschte sie für mehr als zwei Jahre beim Biocon Bristol Myers Squibb Research & Development Center in Bangalore, Indien, bevor sie als Gastwissenschaftlerin im Juni 2021 nach Erlangen kam.
„Ich freue mich sehr über das Stipendium, das nicht nur eine große Ehre für mich ist, sondern mir auch gleichzeitig die Möglichkeit gibt einen neuen und wichtigen Forschungsbereich zu erforschen“, sagt Frau Patel. „Die Gastfreundschaft, mit der ich an der FAU empfangen wurde, ist fantastisch.“
May 2021
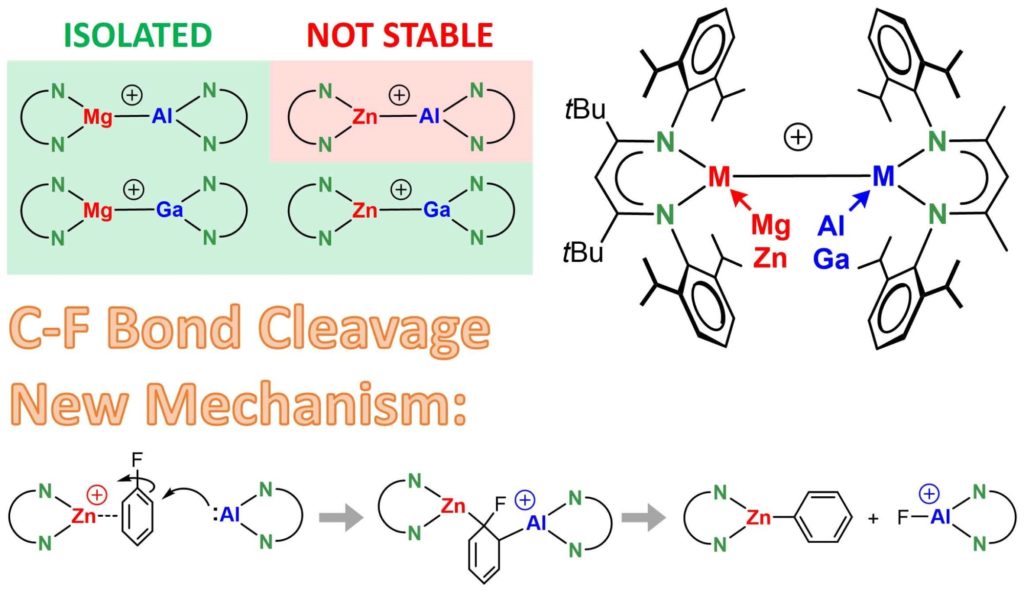
Combining mixing metals with making cations. Our latest contribution by Alexander and Jonathan in Angewandte Chemie.
HETEROBIMETALLIC CATIONS: Mg, Zn, Al, Ga cocktails. Choice of metals strongly influences M-M bond character, stability and reactivity. Their cationic nature is key to facile RT C-F bond cleavage via a new mechanism. (even in p-Me-C6H4-F!) http://bitly.ws/dnZG
May 2021
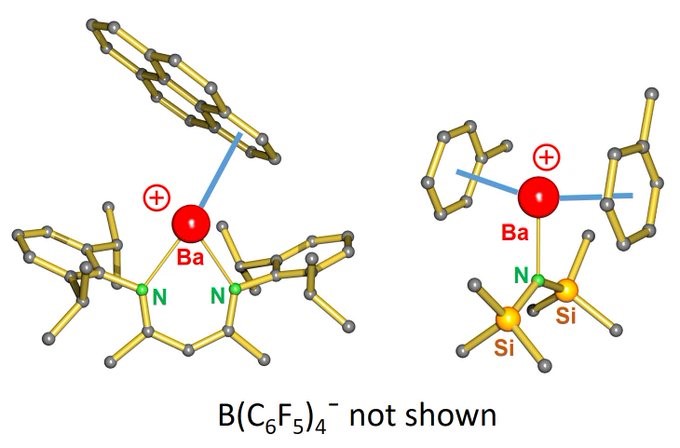
Katharina reports on Lewis base-free cationic complexes of the largest alkaline earth metals Sr and Ba! A BIG challenge to stabilize these BIG metal cations!
“NAKED” STRONTIUM AND BARIUM CATIONS: After Lewis base-free cationic Mg and Ca complexes, we now finally also have the big ones “naked”. (NacNac)Ba+ interacts with a large pi-system. (R2N)Ba+ is sandwiched between toluenes. https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/ej
April 2021
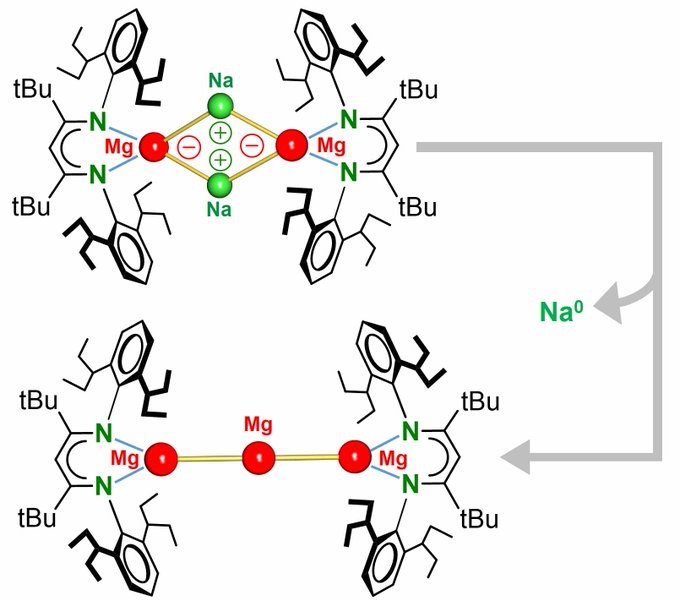
The group made it into NATURE!!! The fantastic cooperative atmosphere within our group enabled isolation of first Mg(0) complexes. Congratulations to all!
ANIONIC MAGNESIUM: Turning R-Mg-I into R-Mg-Na which reacts as a Mg nucleophile RMg(-) Na(+). Heating gave spontaneous Na(+) –> Na(0) reduction (!) and RMgMgMgR, a long sought-after intermediate in Grignard formation. https://nature.com/articles/s4158
March 2021
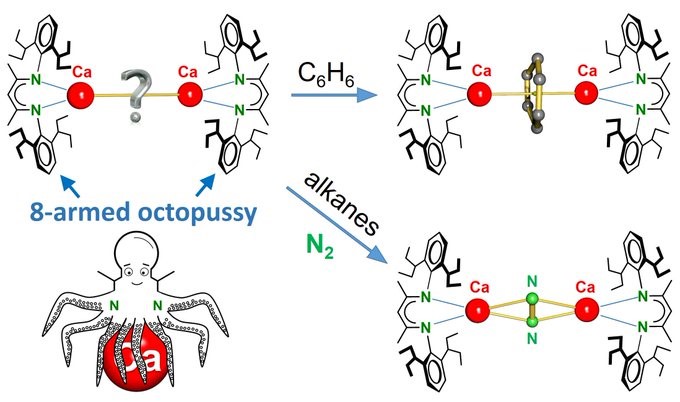
The group made it into SCIENCE!!! It was a hard job to develop this OCTOPUSSY ligand but in the end, hard work always pays off! Congratulations to the chemists who made this giant breakthrough possible!
CRACKING N≡N WITH Ca: Octopussy strikes again! Attempts to isolate a Ca(I) complex gave reduction of aromatic solvents. Thx to many arms complexes are soluble in alkanes. Now N2 is reduced. d-Orbitals on Ca? Is LCaCaL possible? https://bit.ly/3eywhMG
January 2021
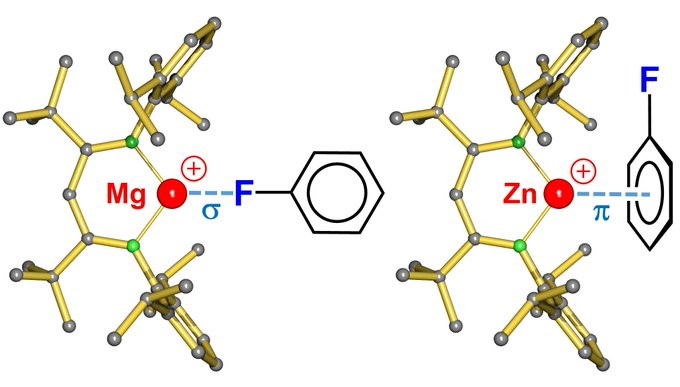
Interesting to see how the two twin metals Mg and Zn can be the same but also so different. Great work from Alexander!
Mg AND Zn: THE SAME BUT DIFFERENT! A first “naked” cationic (BDT)Zn+ complex eagerly forms bonds to arenes and halobenzenes. Despite similar Mg2+ and Zn2+ size, ligand bonding can be quite different due to hard/soft differences. https://pubs.acs.org/doi/10.1021/ac
December 2020
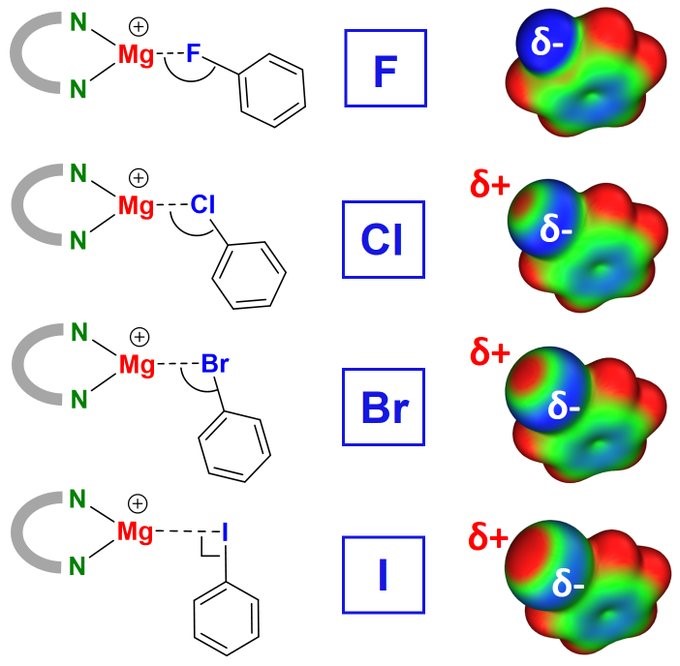
Ever heard of the sigma-hole? Here we show a map. Congratulations Alex and Jonathan on this combined experimental/theoretical project.
Interesting to see MAPPING THE SIGMA-HOLE: Complexation of PhF, PhCl, PhBr, PhI with highly Lewis-acidic Mg cations. Mg…X-Ph angles decrease from F > Cl > Br > I due to the halogen sigma-hole. In all cases there is considerable C-X bond activation. https://pubs.rsc.org/en/content/art
November 2020
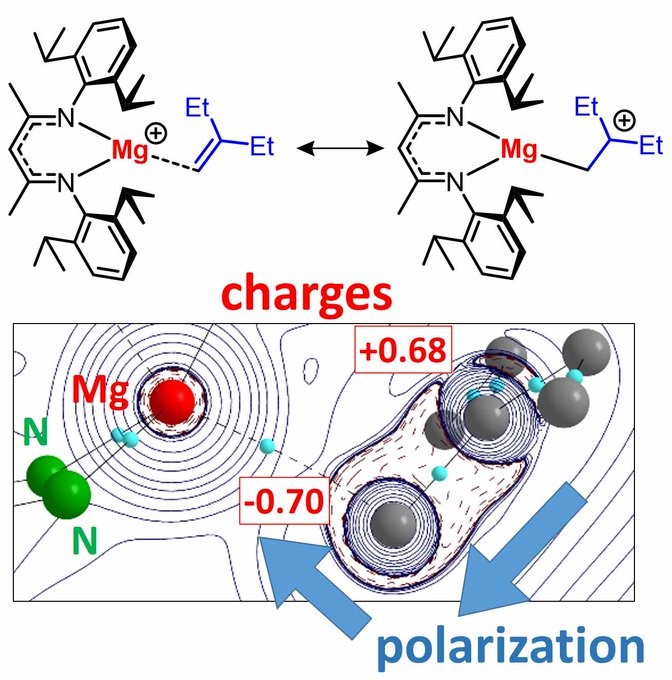
Can main group metals also bind alkenes? Katharina and Alexander say: YES!
UNSUPPORTED MAGNESIUM-ALKENE COORDINATION: Intramolecular s-block metal-alkene complexes are known. We present first INTERMOLECULAR examples. Mg-alkene bonding leads to C=C polarization. Although weak, it is crucial in catalysis. https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/chem.202004716
November 2020
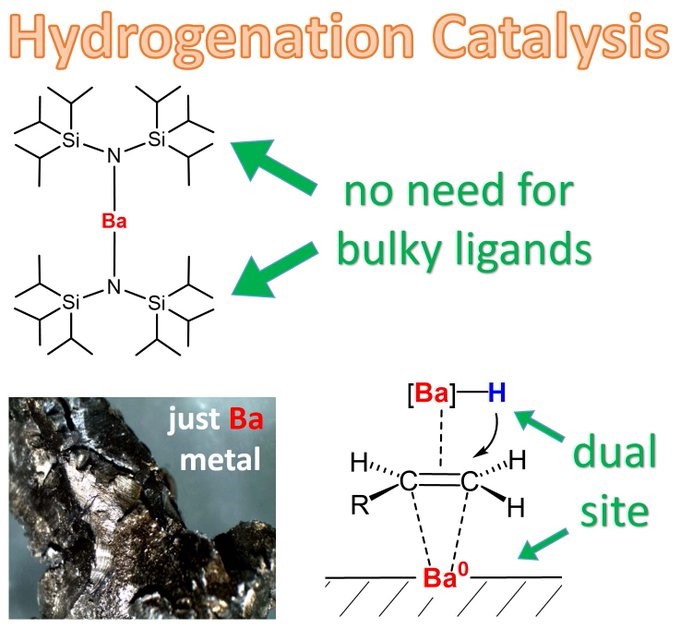
Why using a complicated ligand when just metal is enough? Johannes demonstrates that Ba metal alone is a fantastic hydrogenation catalyst.
NO LIGANDS – JUST METAL: 200 years after Döbereiner’s Pt and 100 years after Sabatier’s Ni, metallic Ba is found to be an all-round hydrogenation catalyst. We propose a DUAL SITE mechanism in which Ba(0) and BaH2 are crucial. https://onlinelibrary.wiley.com/doi/10.1002/an
June 2020


ROYAL SOCIETY MAIN GROUP CHEMISTRY PRIZE
The Boss wins prestigious Award! Or actually the Harder group won. Thanks to all students: past, present, future. https://rsc.org/awards-funding/awards/2020-winners/professor-sjoerd-harder/
June 2020
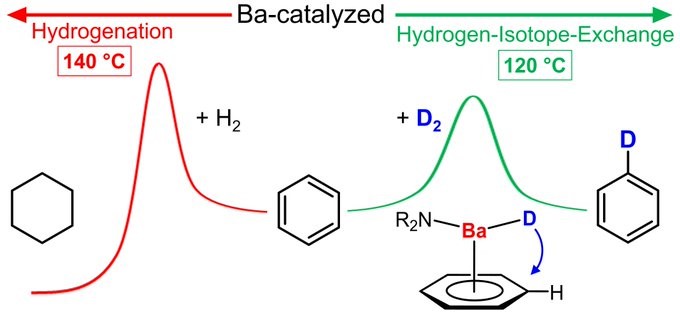
Hydrogen-Deuterium exchange. Johannes shows that you don’t need platinum to this trick. Congratulations with this ACS catalysis paper Johannes!
IS BARIUM THE NEW PLATINUM? Recently we showed Ba catalysts for benzene hydrogenation. At slightly lower T fast Hydrogen-Isotope-Exchange is observed. TOF’s > 250 h-1 are comparable to noble metal activities. Key step = nucleophilic aromatic substitution. https://pubs.acs.org/doi/10.1021/ac
May 2020
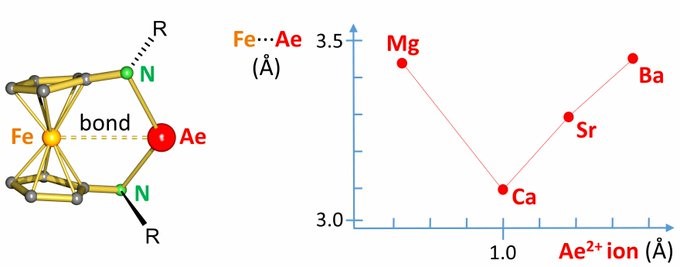
Are d-orbitals on Ca, Sr and Ba really important???? We slowly start to think that Ca, Sr and Ba should be part of the d-block. Congratulations with this Angewandte Chemie paper Phil!
D-ORBITALS FOR CALCIUM? Fe-Ae bonding is observed for Ae = Ca, Sr and Ba but not for Mg. Bonding involves vacant d-orbitals on Ca, Sr or Ba and partially filled d-orbitals on Fe. Joint paper with Siemeling, Frenking & Sarkar. http://doi.wiley.com/10.1002/anie.202005774
May 2020
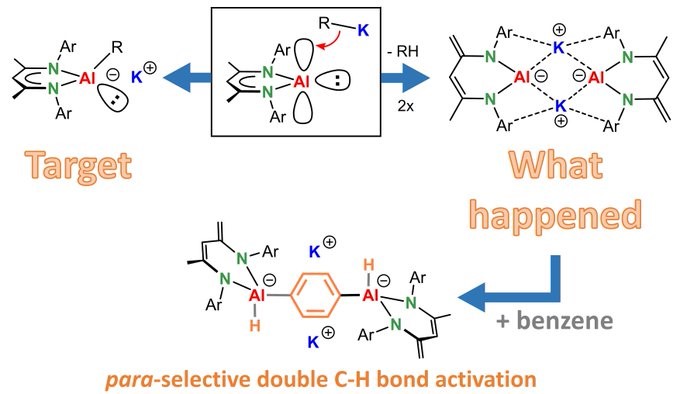
Not exactly what we wanted but still highly interesting. Our first contribution to the world of anionic Al complexes. Congrats Sam!
BOOSTING THE REACTIVITY OF LOW-VALENT AL(I): Not really what we wanted but what we got is certainly just as reactive. Highly para-selective C-H activation of benzene via a template effect. https://t.co/T3xvgTFwwT?amp=1
March 2020

So glad this book is out!!! Editing and for a large part writing it was A LOT OF WORK! But the result is a great book that feeds the young generation (hopefully) for the next 10 years.
Februar 2020
We hit the Angewandte Chemie again with a hydrogenation paper. Quite amazing how these very bulky amide ligands boost the activity of the catalyst. We like BIG!!!
BARIUM – THE NEW NICKEL? Superbulky barium amides are champion hydrogenation catalysts. Not only for challenging alkenes but even for arenes including highly stable aromatic benzene! I think we got a NEW FAVORITE METAL! https://onlinelibrary.wiley.com/doi/10.1002/an
August 2019
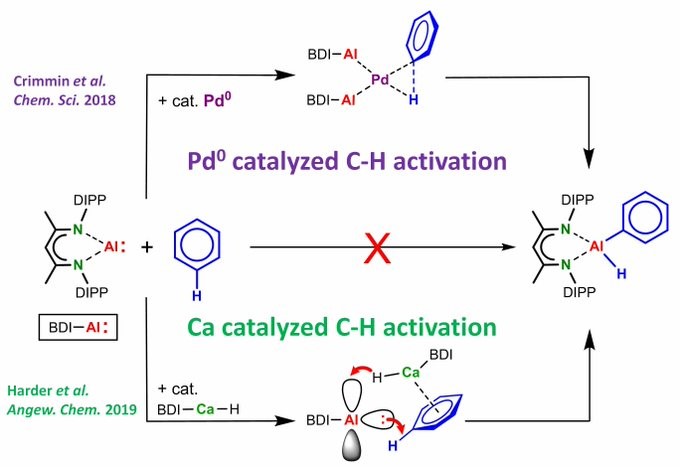
People always believe you need transition metals for C-H bond activation…. Here we demonstrate that a combination of Al and Ca also works absolutely fine! Congratulations Steffen to this HOT Angewandte Chemie paper!
C-H BOND ACTIVATION WITHOUT Pd: (BDI)Al does not react with benzene. Addition of a Ca hydride catalyst gave smooth C-H bond activation at room temperature. Hydride –> Al attack makes Al anionic boosting its nucleophilicity ! https://doi.org/10.1002/anie.201908978
August 2019
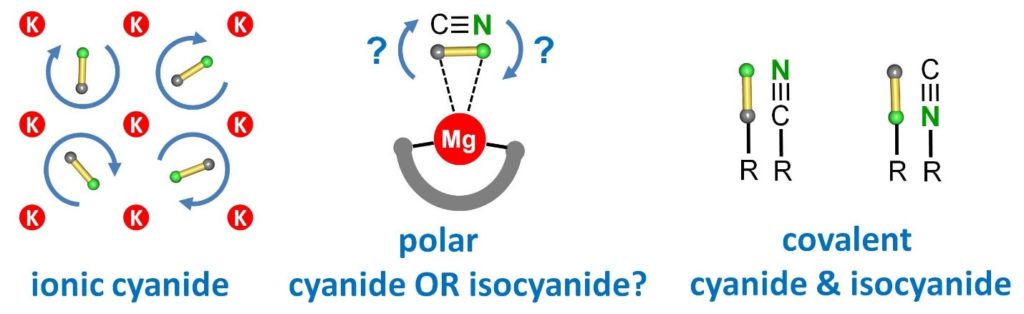
How does a cyanide bind to magnesium? Via C or N? The answer is: BOTH! Great work from Gerd now published in Angewandte Chemie!
CYANIDE OR ISOCYANIDE? CN- ions in ionic KCN rotate fast and behave as pseudohalogens. Covalent CN compounds are either cyanides or isocyanides. What about polar Mg-CN complexes? See our latest contribution: https://onlinelibrary.wiley.com/doi/10.1002/an
May 2019
This is our contribution to the YEAR OF THE PERIODIC TABLE.
We put Calcium on the map!
https://chemistrycommunity.nature.com/posts/49408-ca-for-ca-talysis
https://nature.com/immersive/d42859-019-00001-7/index.htm
February 2019
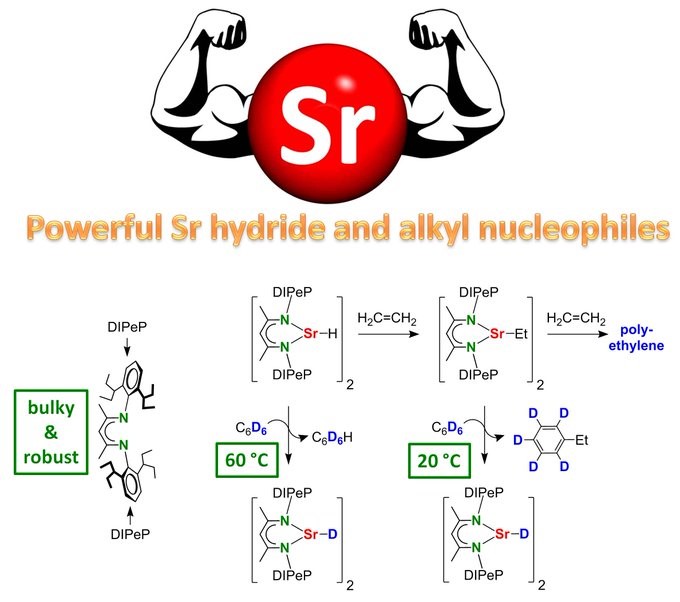
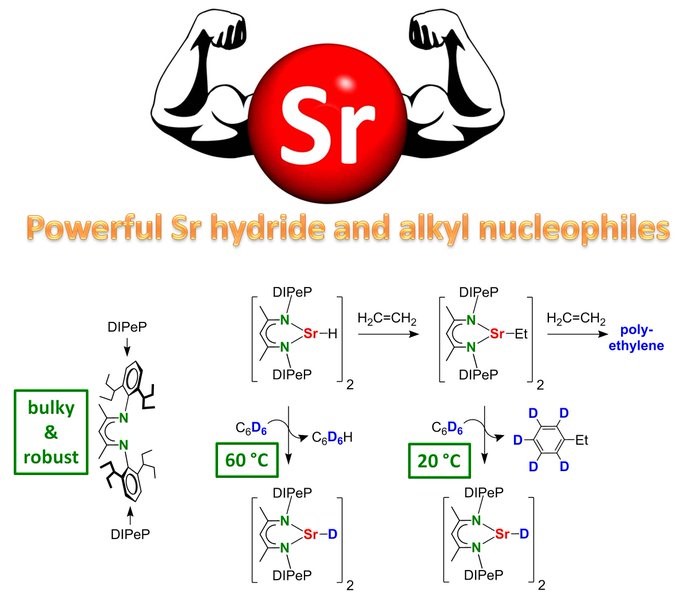
Bastian made the very first real (unstabilized) Strontium alkyls. Oh boy, they are reactive!!! Nucleophilic addition to benzene & first alkaline-earth metal catalyzed benzene alkylation! Great work Bastian, another Angewandte Chemie paper!
BIG BROTHER ALKYL STRONTIUM IS BORN. The key is the superbulky DIPeP ligand. Alkyl/H exchange with benzene proceeds already at room temperature! Even the Sr hydride complex reacts with benzene thus enabling catalytic deuteration of C6H6 with D2: https://onlinelibrary.wiley.com/doi/10.1002/an
January 2018
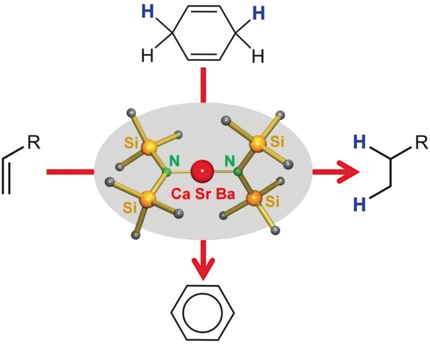
Katharina and Heiko show that transfer hydrogenation can also be done with alkaline-earth metal catalysts instead of with precious transition metals. A well-deserved Angewandte Chemie paper!
TRANSFER HYDROGENATION WITH CALCIUM: Simple Ca(NR2)2 catalysts effectively transfer hydrogens from 1,4-cyclohexadiene (a waste product from plant oil refinery) to alkenes. Catalysis with big brother Ba is fast hydrogenating styrene at room temperature. https://onlinelibrary.wiley.com/doi/10.1002/an
November 2018
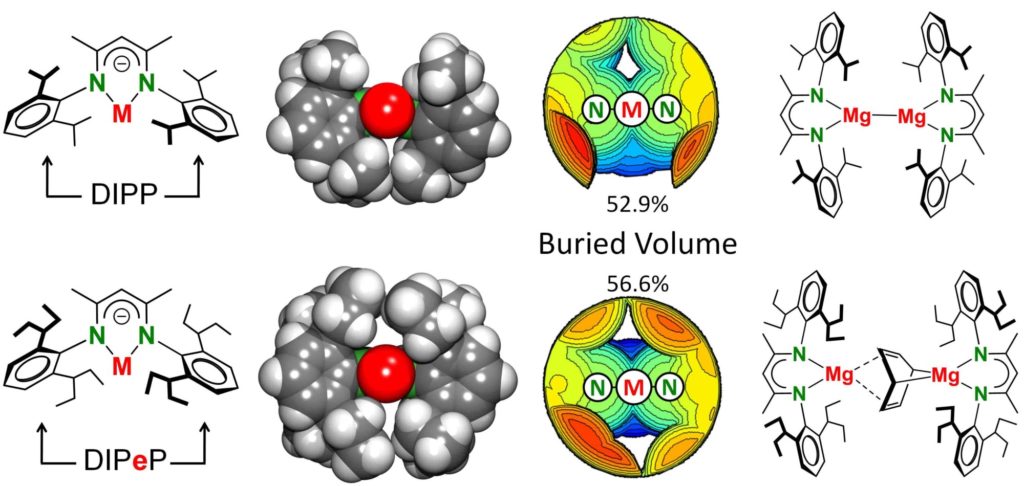
Big ligands can do everything! Thomas made this big boy for the first time and together with Mg it can reduce benzene to anti-aromatic C6H6(2-)! Again an Angewandte Chemie paper
SUPERBULKY NACNAC: iso-pentyl may not be that much bulkier than iso-propyl but DIPeP-nacnac certainly PEPs up chemistry. Attempts to isolate (DIPeP-nacnac)Mg• led to reduction of the aromatic benzene solvent to give anti-aromatic C6H6(2-). https://onlinelibrary.wiley.com/doi/10.1002/an
September 2018
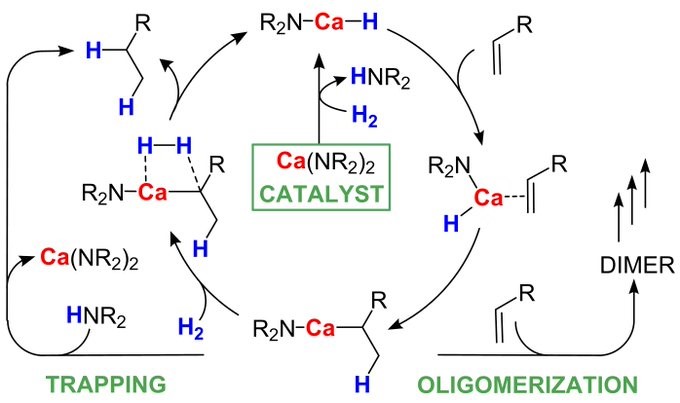
Great follow-up paper on our publication in Nature Catalysis. Never thought it would be that simple! Ca catalyzed alkene hydrogenation with Ca[N(TMS)2]2. Expected the HN(TMS)2 to be detrimental for any intermediate. Turns out it is the excellent trapping agent that prevents polymerization! With Ba it is flying! https://onlinelibrary.wiley.com/doi/epdf/10.10
August 2018
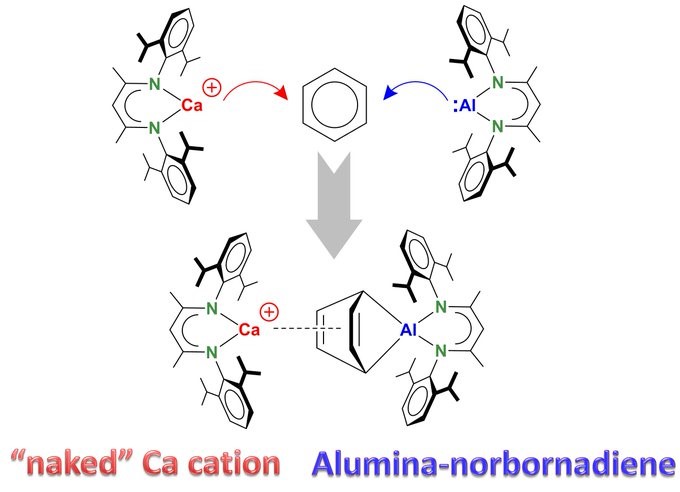
Congratulations to another Angewandte Chemie paper Steffen!
DEAROMATIZATION OF BENZENE – by combined attack of a „naked“ highly Lewis acidic cationic Ca complex and a nucleophilic Al complex. The product resembles an alumina-norbornadiene coordinating to Ca. The mechanism is likely of the FLP type. https://onlinelibrary.wiley.com/doi/abs/10.100
Juli 2018
Jürgens and Steffens work about naked cationic magnesium and calcium complexes has been chosen as cover picture on the new Chemical Communications issue. Great work!
Benjamin Freitag finished his PhD degree with a successful defense. Congratulations Ben and all the best for your future!
Juni 2018
Jonathan Eyselein changed flasks for computers and stays at the theoretical chemistry department for 6 months until he comes back to ou group for his PhD thesis.
April 2018
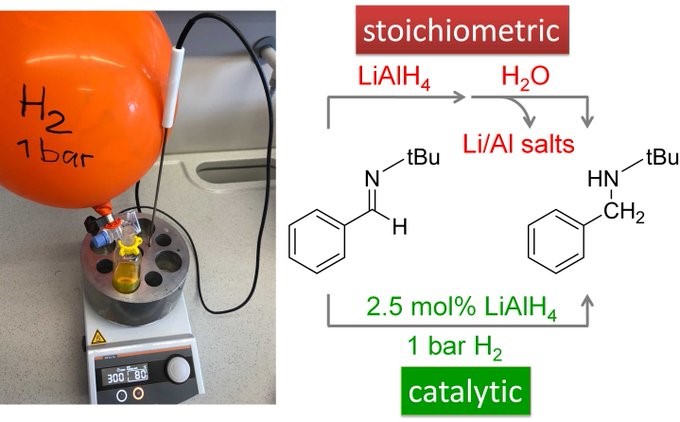
Would you have ever though that LiAlH4 is not just a stoichiometric reagent but can also be a catalyst? First observations by Christian have been worked out by Holger. Congratulations to this fine Angewandte Chemie paper!
GREEN CHEMISTRY WITH ORANGE BALLOONS: Perform the classical LiAlH4 reduction of imines under H2 instead of under N2 and you save yourselves a lot of LiAlH4 reagent, tedious work-up and salty Li/Al byproducts! See our latest article: https://doi.org/10.1002/ange.2
April 2018
Jonathan Eyselein finishes his Master Thesis in our group. Congratulations, Joni!
Christian Knüpfer starts with his Specialization Module and will continue afterwards with his Master Thesis. Welcome in our group!
January 2018
Very proud of the group to contribute to the very first issue of Nature Catalysis – the leading catalysis journal! Imine hydrogenation with simple alkaline earth metal catalysts
nature.com/articles/s41929-017-0006-0
December 2017
Bernhard Auer finished his master thesis and will move to New Zeeland for his PhD work. Enjoy your time at the other side of the earth!
November 2017
Andrea Causero defended his PhD thesis as the final step to achieve his PhD degree. Congratulations Andrea and all the best for your future career!
August 2017

Andreas Rösch finished bis master thesis in our group and will join Prof. R. Kretschmer at the University of Regensburg. All the best, Andi!
Michael Wiesinger got awarded with the poster price of the AGIChem conference (Göttingen) for his poster about alkaline earth metal hydride complexes. Congratulations Michael, well done!
June 2017

Dr. Brant Maitland leaves our group. All the best in the future!
Christian Fischer was sponsored by the Graduate School Molecular Science (GSMS) of the FAU Erlangen to join the ICHAC-12 conference in Vancouver (Canada). His poster about bora-amidinate supported group 2 metal catalysis was awarded with the poster price. Congratulations Christian!
April 2017
Samuel Grams and Michael Wiesinger join your group as PhD students. Welcome to the Harder Group!
February 2017
Dr. Ana Melissa Escalona Bauza joined our group.
Samuel Grahms and Michael Wiesinger finished their Master Theses in the Harder Group. Congratulations!
November 2016
Alexander Friedrich and Bastian Rösch finished their Master Theses and will stay in the group as PhD students.
Oktober 2016
Katharina Thum finished her Master Thesis and joines the group as a PhD student.
Dr. Carlos Dücker-Benfer leaves our group to work in the safety department of the FAU.
January 2016
Steffen Brand, Holger Elsen, Johannes Martin and Philipp Stegner joined the group as PhD students
Oktober 2015

Prof. Sjoerd Harder held his inaugural lecture
March 2015


Dr. Brant Maitland joined the group as post-doc.
The new orange NMR magnets arrived and are being built up.
Also new orange chairs arrived.
February 2015
Gerd Ballmann joined the group as PhD student.
January 2015
Two new members joined the group:
Thomas Gentner as PhD student
Christian Fischer as PhD Student
November 2014
Dr. Jens Langer joined the group
July 2014
Dr. Heiko Bauer joined the group as post-doc.
April 2014
Dr. Tom Stennett joined the group as post-doc.
September 2013
Two guests joined the group:
- Magnus Buchner just finished a postdoctoral research project at the University of York
- Brant Maitland is a PhD student from the Cameron Jones group at Monash (Australia) and swapped southern spring for northern autumn to work here on bulky nacnacs.
August 2013

Our brilliantly ORANGE luminescent superbulky europocene made a nice front cover for Chemistry European Journal.
Check it out! http://onlinelibrary.wiley.com/doi/10.1002/chem.201390139/abstract
July 2013
Markus Jurisch joined the group as a post-doc.
April 2013
Andrea Causero joined the group as a PhD student.
March 2013
Wu Xian joined the group as a post-doc.
February 2013
Benjamin Freitag joined the group as a PhD student.